 
The other neutrons either escape out or are absorbed in any other medium except uranium. It is possible to produce such conditions in which only or neutron, out of all the neutrons created in one fission reaction becomes the cause of further fission reaction. In this way, this process rapidly proceeds and in an infinitesimally small time a large amount of energy along with huge explosion produces, it is a representation of fission chain reaction If conditions are appropriate these neutrons produce fission in some more atoms of uranium. Out of this reaction, about three neutrons are emitted. Suppose that we have a definite amount of and a slow neutron originating from any source produces fission reaction in one atom of uranium. The process is called a fission chain reaction. By properly using these neutrons fission reaction can be produced in more uranium atoms such that a fission reaction can continuously maintain itself. We have observed that during fission reaction a nucleus of uranium-235 absorbs a neutron and breaks into two nuclei of almost equal masses besides emitting two or three neutrons. However, it not been observed that fission takes place very easily with the slow neutrons in uranium-235 and plutonium-239, and mostly these two are used for fission purposes. Fission reaction is not confined to uranium along it is possible in many other heavy elements. All of these products (fragments) are radioactive. 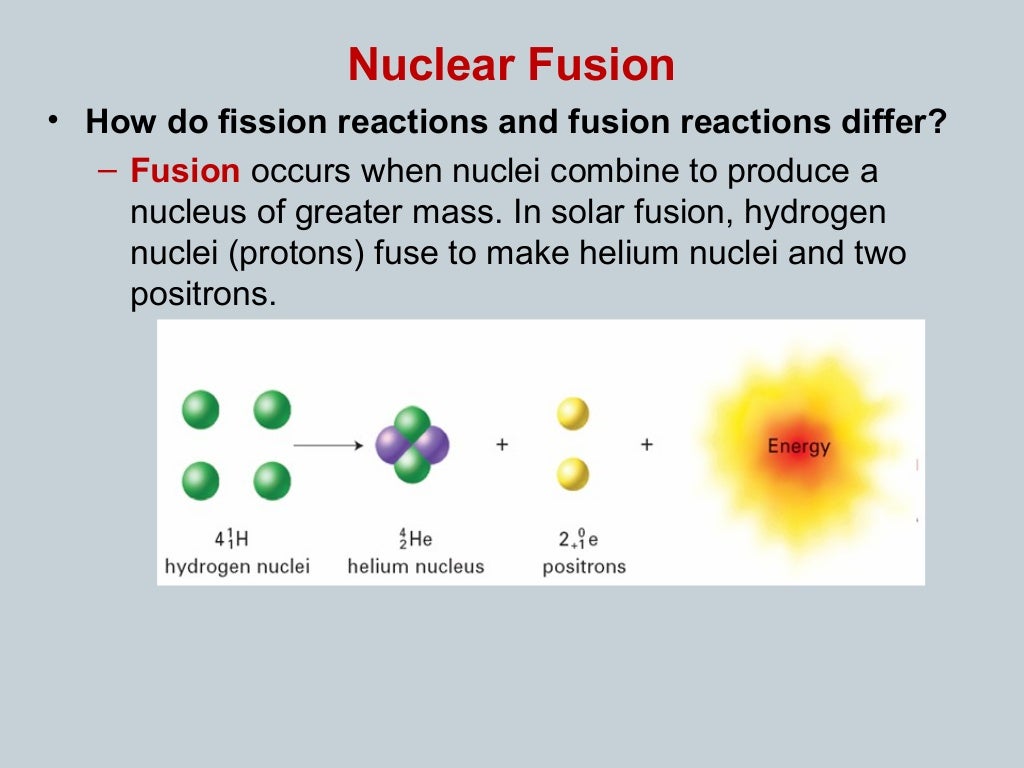
Hence in the uranium fission reaction, several products may be produced. Two possible fission reactions of uranium are given below as an example: In fact, any of the two nuclei present the upper Horizontal part of binding energy that could be produced. The fission process of uranium does not always produce the same fragments (Ba, Kr). This means that an energy 235 × 0.9 = 211.5 MeV is given out in the fission of one uranium nucleus. 
Thus when a uranium nucleus breaks up, as a result of fission reaction, into barium and krypton, then energy at the rate of 0.9 MeV per nucleon is given out. This graph shows that the binding energy per nucleon is greatest for the middle elements of the periodic table and this bending energy per nucleon is a little less for the light or very heavy elements i.e., then nucleons in the light or very heavy elements are not so rigidly bound.įor example, the binding energy per nucleon for uranium is about 7.7 MeV and the products of the fission reaction of uranium, namely barium and krypton, have a total mass less than the mass of uranium equal to 8.5 -7.6 = 0.9 MeV per nucleon. It may be kept in mind that three is no difference between the sum of the mass and the charge number on both sides of the equation.įission reaction can be easily explained with the help of a graph. Here Q is the energy give out in this reaction, By comparing the total energy on the left side of the equation with total energy on the right side, we find that in the fission of one uranium nucleus about 200 MeV energy is given out. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
